Wearables Keep Creeping from Wellness Toy to Trial-Grade Endpoint Collection
Mar 22, 2026
A biotech is using Apple Watch data in a Parkinson's drug study. The headline is modest; the implications are not. Here is what the evidence actually says about wearables in clinical trials, and why this moment matters more than it looks.
Most Parkinson's drug trials still measure patients the same way they did twenty years ago. A participant comes into the clinic every few months. A neurologist runs a structured assessment. The score gets recorded. The patient goes home.
The problem is that Parkinson's doesn't cooperate with that schedule. Tremor burden, gait quality, and motor fluctuations shift hour by hour and day by day. A clinic visit captures one snapshot of a disease defined by variation. That makes it hard to detect small but real treatment effects, especially early in the disease course when changes are subtle.
That is the gap pharma is starting to try to close with wearables.
One Biotech, One Apple Watch, One Trial
STAT News reported in March 2026 that a biotech is using Apple Watch data in a clinical study of a Parkinson's drug. The headline sounds modest. The implications are not.
The question it raises is not whether Apple makes medical-grade hardware. It doesn't, at least not in the full regulatory sense. The question is whether commercially scaled consumer devices, with the right analytics and protocols wrapped around them, can generate data credible enough to support endpoint collection in a regulated drug study.
If the answer turns out to be yes, even partially, it changes how sponsors design trials for diseases that involve continuous, fluctuating symptoms.
Why Parkinson's Is the Right Test Case
Not every disease suits wearable sensing. Parkinson's is a strong candidate for several reasons.
The core symptoms are continuous and observable: tremor, bradykinesia (slowed movement), gait instability, and motor fluctuations tied to medication timing. These manifest as movement patterns that accelerometers and gyroscopes can detect. Unlike lipid levels or tumor markers, Parkinson's symptoms don't require a blood draw or imaging scan to observe. They're present at home, at work, during sleep, during a walk across the room.
Clinic visits miss most of this. A 30-minute exam captures a narrow slice of a patient's daily motor experience. If a drug reduces average tremor by 15% across the day but the patient happens to be having a good two hours at exam time, that effect won't show up in the chart. Continuous monitoring doesn't have this problem.

What the Evidence Actually Says
This isn't speculative. Peer-reviewed work already supports the concept.
Researchers in the WATCH-PD study, published in npj Parkinson's Disease in 2024, followed patients with early, untreated Parkinson's disease for 12 months using commercially available smartwatch and smartphone sensors. They tracked gait, tremor, and related movement features continuously over that period. The study found significant longitudinal change in these measures over 12 months, and the authors concluded the approach shows promise for assessing therapeutic efficacy in early Parkinson's disease.
That is a peer-reviewed longitudinal study using consumer hardware and detecting treatment-relevant signal. That matters.
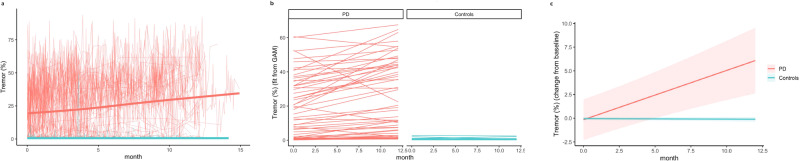
Regulatory acceptance has followed the evidence in at least one case. In 2022, Rune Labs received FDA clearance for StrivePD, an Apple Watch-based tool for tracking Parkinson's symptom patterns. The FDA cleared it for clinical monitoring use. That is a formal acknowledgment that watch-derived symptom data can play a role in clinical contexts, not just consumer wellness apps.
By 2023, STAT News was already covering multiple Apple Watch apps cleared for Parkinson's monitoring, noting the category was becoming more clinically meaningful. The direction of travel has been consistent.
What Still Has to Be Proven
None of this means consumer wearables are ready to serve as primary endpoints across drug development. They aren't. Several hard problems remain.
- Validation: A measure that tracks longitudinal change doesn't automatically become a validated clinical endpoint. That requires additional work: bridging studies, regulatory qualification, and consensus on what constitutes a clinically meaningful change in wearable-derived scores.
- Regulatory acceptance: The FDA cleared StrivePD for symptom monitoring, not as a primary efficacy endpoint in a pivotal trial. Exploratory and secondary endpoints are where this technology lives right now, not first-line registration endpoints in most cases.
- Data quality: Wearables generate messy real-world data. Inconsistent wear time, firmware variation across device generations, missing data from charging gaps, and signal artifacts from environmental motion all complicate analysis. Clinical trials need robust protocols to handle this.
- Disease fit: Parkinson's movement symptoms happen to be well suited to accelerometer-based sensing. Many other conditions are not. Extrapolating from this success case to other therapeutic areas requires caution.
What Changes If This Works
If wearable-derived measures become accepted as valid secondary or exploratory endpoints in Parkinson's trials, the downstream effects on study design are significant.
Sponsors get denser data. Instead of quarterly site visits, they get daily data streams. That provides more statistical power to detect small effects and more information about when and how treatment works across the course of a day.
Trials become less site-dependent. If participants can generate valid endpoint-adjacent data at home, studies can include people who live far from academic medical centers or face mobility limitations that make frequent travel difficult. That changes who can participate.
Responders may be identifiable faster. Continuous monitoring makes it easier to see who is improving and who is not, potentially accelerating adaptive trial designs and informing dose-finding decisions with more granularity than episodic clinic visits allow.
None of this replaces the clinic. Drug approval still requires safety data, physician-rated outcomes, and formal clinical assessments. Wearables are supplementing the evidence stack, not replacing it.
The Molecular Picture Still Matters
As behavioral endpoints from wearables gain traction in neurological trials, the molecular side of the evidence stack doesn't shrink. It becomes more important.
Wearable data tells you how a patient is moving. It doesn't tell you what is happening at the protein level. For Parkinson's drug development, that means CSF biomarkers such as alpha-synuclein aggregation state, phosphorylated tau, and inflammatory markers still need to be measured from biological samples. Behavioral and molecular data are complementary, not substitutes for each other.
The Bigger Shift
The Apple Watch story isn't really about Apple. It's about a deeper change in what sponsors want from clinical measurement.
The industry has been trying for years to move from episodic, clinic-based snapshots toward continuous, patient-level data streams. Wearables are one pathway to that goal. They aren't the only one, and they aren't mature enough yet to anchor most trial designs. But in a disease like Parkinson's, where movement symptoms are the target, continuous sensing is closer to right than intermittent clinic visits ever were.
Whether consumer wearables eventually clear the bar for primary endpoints in pivotal trials remains to be seen. The regulatory path is long and the evidence requirements are high. But the question pharma is starting to ask has changed. It's no longer whether consumer devices can generate useful clinical data. It's which devices, in which diseases, with which analytical frameworks, and at what level of evidence maturity they can start being used.
That is a harder question than the headline suggests. It's also a more interesting one.
References
- Adams JL, et al. (2024). Using a smartwatch and smartphone to assess early Parkinson's disease in the WATCH-PD study over 12 months. npj Parkinson's Disease. doi.org/10.1038/s41531-024-00721-2
- Rune Labs' Apple Watch-based Parkinson's disease tracker gets FDA clearance. The Verge. June 2022. theverge.com
- For Parkinson's disease, advances spurred by Apple Watch offer a glimmer of hope. STAT News. October 2023. statnews.com
Have questions about this research or our products?
Contact Our TeamGet new posts delivered to your inbox