A Human Uterus Survived Outside the Body for 24 Hours. Here's Why That Matters.
Mar 29, 2026
A team in Valencia recently kept a donated human uterus viable for 24 hours using an ex vivo perfusion machine. The real story isn't about artificial wombs. It's about what this kind of organ-engineering could mean for uterus transplantation logistics and reproductive biology research.
A donated human uterus connected to a perfusion machine, circulating modified blood at body temperature, staying metabolically active for roughly 24 hours. That's the result reported in March 2026 by a team at the Carlos Simon Foundation in Valencia, Spain. The work hasn't been published yet. The machine is called "Mother." The approach borrows directly from organ transplantation engineering.
The coverage has focused on the most speculative angle: whether this technology could eventually support external gestation. That question isn't what makes the work interesting. The nearer-term implications are more grounded, and more useful.
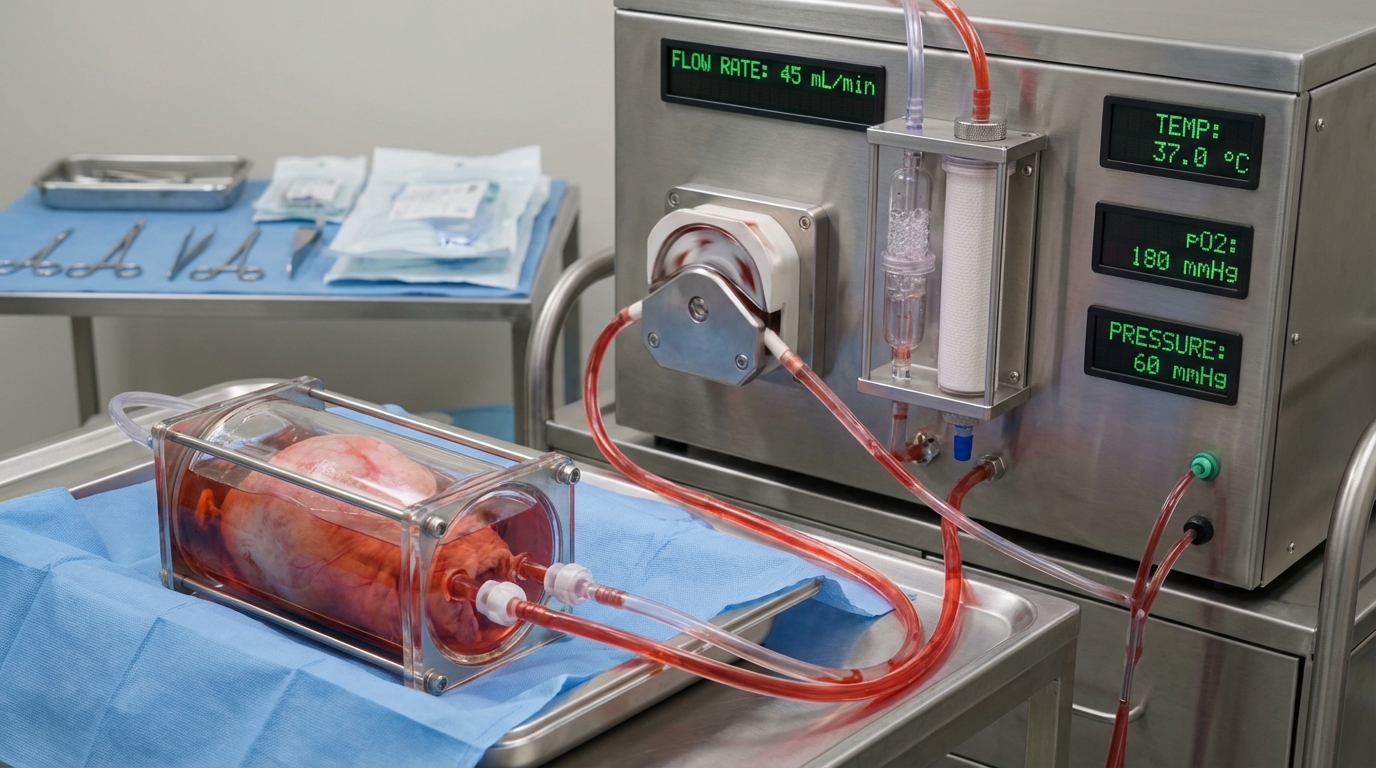
Machine Perfusion Is Already Serious Science
Normothermic machine perfusion isn't new. In liver, kidney, and heart transplantation, perfusion systems have been advancing for more than a decade. The core idea is straightforward: circulating warm, oxygenated, nutrient-rich fluid through an explanted organ maintains viability better than static cold storage, reduces ischemic injury, and creates a window for assessment.
In 2025, Drivas and colleagues published a comprehensive review of ex vivo machine perfusion in uterus transplantation (Transplant International, 2025), covering 31 studies. Their conclusion was that EVMP is an active and growing research strategy in this field, with both preservation and translational applications.
What the Carlos Simon team did is an extension of that trajectory into human tissue, for the first time.
The Uterus Transplant Problem
The first live birth from a transplanted uterus occurred in 2014 in Sweden. By 2025, uterus transplant programs operated on multiple continents. The limiting factor isn't surgical skill. It's time.
Deceased-donor uteri are constrained by warm and cold ischemia. The window between organ recovery and reperfusion narrows quickly, and every minute of ischemic injury affects outcomes. Uterus transplant reviews have flagged this as a significant practical barrier to expanding deceased-donor programs.
A 2024 preclinical study (Loiseau et al., Transplantation Direct) addressed this directly, testing hypothermic machine perfusion against static cold storage in a porcine model. The machine perfusion group showed improved reperfusion capacity, better tissue oxygenation, and more favorable ATP levels during the preservation phase. Histological sections confirmed less tissue injury.
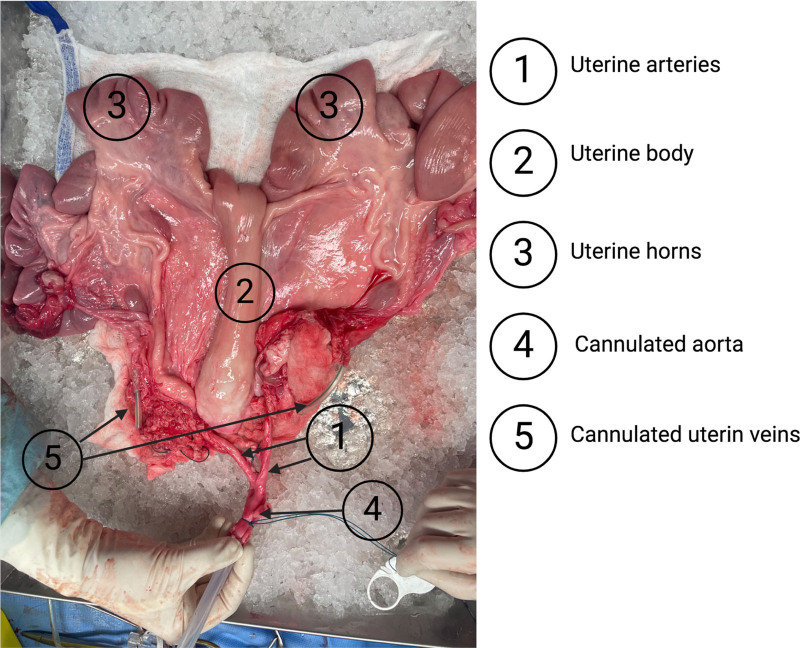
A Platform for Studying Uterine Biology
The transplant logistics angle is real. But there's a second reason an ex vivo uterus system matters: research access.
The uterus is surprisingly hard to study. Endometrial biopsy gives a snapshot. Cell culture loses three-dimensional tissue architecture and the cellular crosstalk that governs receptivity. Animal models don't fully recapitulate human reproductive biology. In vivo human studies are expensive, slow, and ethically complex.
Implantation failure is one of the least-understood bottlenecks in reproductive medicine. Most IVF cycles that fail do so at or before the embryo attaches. Understanding what the endometrium does during the receptive window, in living tissue, under controlled conditions, is exactly what a functioning ex vivo model could enable. The same applies to fibroids, endometriosis, and menstrual-cycle biology. These are common conditions where the basic science is still limited by access to viable human tissue.
Research Matrices for Reproductive Biology
Labs working on uterine biology, implantation markers, and reproductive endocrinology need matrices that reflect the biology they're measuring. Assay validation for prenatal and fertility biomarkers requires human serum from donors in relevant physiological states.

Available as pooled or individual donor samples across all three trimesters, this product supports assay development for the kinds of reproductive biomarkers that a living ex vivo uterus system could eventually help validate in functioning tissue.
What Remains Speculative
The team's founder has described a longer-term vision: whether this platform could someday support partial or full gestation outside the body. That ambition is clearly stated in the MIT Technology Review coverage. It is also clearly speculative.
A 24-hour proof of concept in one donated organ is separated from gestation support by engineering challenges that don't yet exist in the literature. No published work demonstrates anything close to that capability. The current result should be read as what it is: a first step establishing that sustained ex vivo uterine perfusion is possible in human tissue.
That's enough to be significant.
The Bigger Picture
Reproductive medicine has made enormous strides through molecular biology and genetics. But the uterus has remained relatively opaque as a research object, partly because of limited access to functional human tissue outside the body. An ex vivo perfusion system changes that ratio, at least for the duration of its viability window.
Better transplant logistics and better research access are the near-term value here. The further speculative possibilities may or may not prove out. The field will need published data, reproducible methods, and peer review before anyone should treat this as anything more than a promising first step. Watch for the paper.
References
- MIT Technology Review. "A woman's uterus has been kept alive outside the body for the first time." March 28, 2026. technologyreview.com
- Drivas EM et al. A Comprehensive Review of Ex-Vivo Machine Perfusion in Uterus Transplantation. Transplant International, 2025. PMID: 41439262
- Loiseau E et al. Uterine Transplant Optimization From a Preclinical Donor Model With Controlled Cardiocirculatory Arrest. Transplantation Direct, 11(1), 2024. DOI: 10.1097/TXD.0000000000001735. PMC11634325
- Past, Present, and Future: A Review of Uterus Transplant. Transplant International, 2025. PMC12702789
Have questions about this research or our products?
Contact Our TeamGet new posts delivered to your inbox