What Medicine Misses When It Ignores the Body's Energy Budget
Mar 15, 2026
Biomedicine excels at mapping molecules. But organisms run under finite energy budgets, and diseases involving aging, neurodegeneration, and chronic inflammation may be better understood when framed partly as competition over those resources.
Biomedicine has gotten very good at mapping parts: gene sequences, protein structures, receptor binding affinities, signaling pathways that branch into further pathways. The field has been extraordinarily productive at this level of analysis, and the clinical payoffs are real.
But a Nature News & Views essay published this spring asks a different kind of question. What if we've been systematically under-modeling the fuel bill?
The Core Argument
The thesis is not that molecular biology has failed. It's that organisms operate under finite energy budgets, and disease may be better understood when framed partly as competition over those resources, not instead of molecular mechanisms, but in addition to them.
This builds on something well-established in evolutionary biology and metabolic ecology. Herman Pontzer's work on constrained total energy expenditure found that the body doesn't simply scale energy output with demand. Instead, it compensates by reducing allocation elsewhere. His 2021 Science study tracking 6,421 people across the lifespan found that energy expenditure follows predictable biological constraints at every life stage. Brown and colleagues' Metabolic Theory of Ecology extended the principle further: energy constraints govern growth rates, reproduction, lifespan, and body size across organisms, and they do so more universally than most molecular mechanisms.
That cross-species reliability is the key point. Molecular pathways fragment by species, tissue, and context. Physical energy constraints are governed by thermodynamics. They generalize better, which may partly explain why so many promising mouse findings don't translate to humans: the specific molecules differ, but the energy arithmetic doesn't.
Where the Evidence Already Points
Several subfields have been working with this framing, often without labeling it as such.
Sickness behavior as energy reallocation
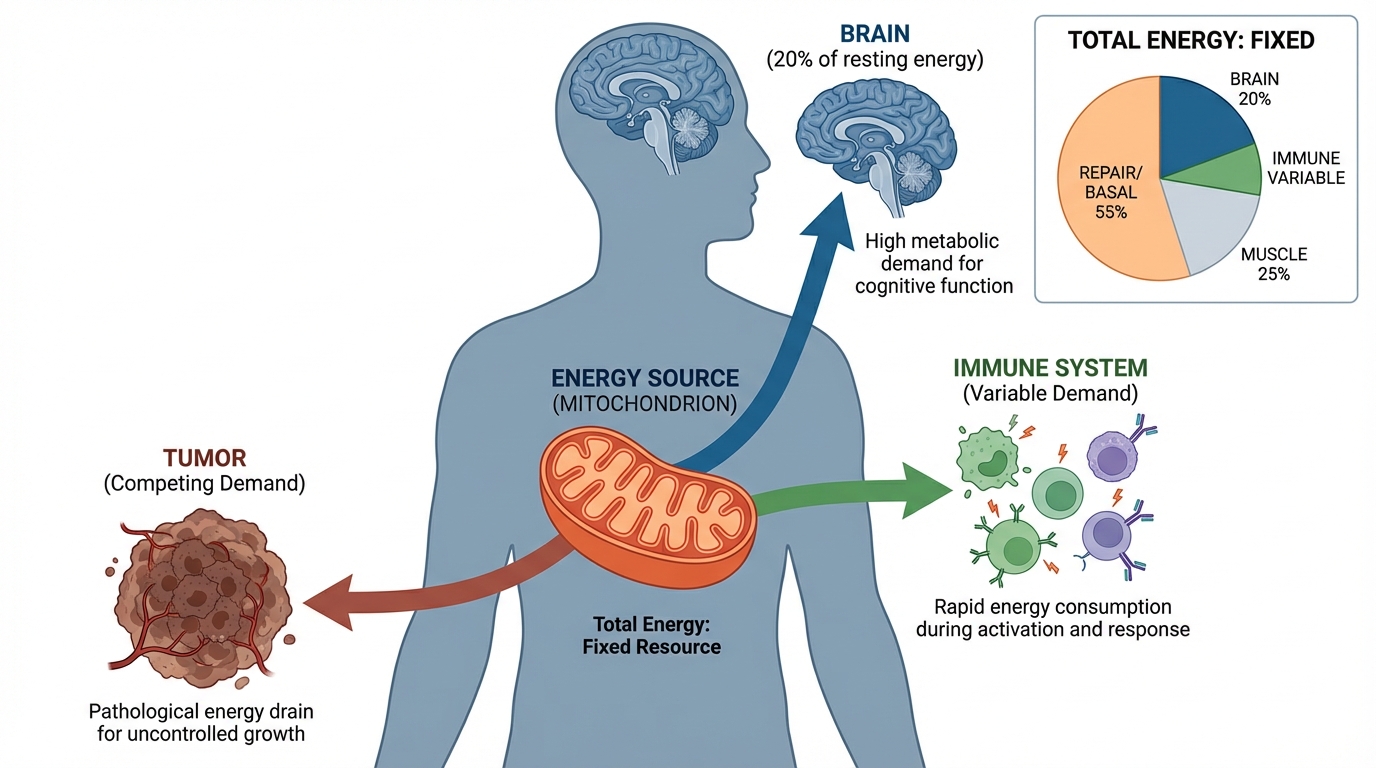
When the immune system activates, it costs energy. Wang et al. (2024) reviewed how systemic infection triggers a coordinated reallocation in Cellular and Molecular Immunology: appetite drops, mobility decreases, social withdrawal occurs, and mood changes. These aren't incidental side effects of inflammation. They look like deliberate constraints imposed by an organism running near its energy ceiling. The body reduces discretionary spending to fund the immune response.
The parallel in clinical medicine is underappreciated. Treatment-related fatigue, appetite loss, and behavioral changes during intensive therapy may not be purely toxicity. They may reflect the same kind of budget reallocation.
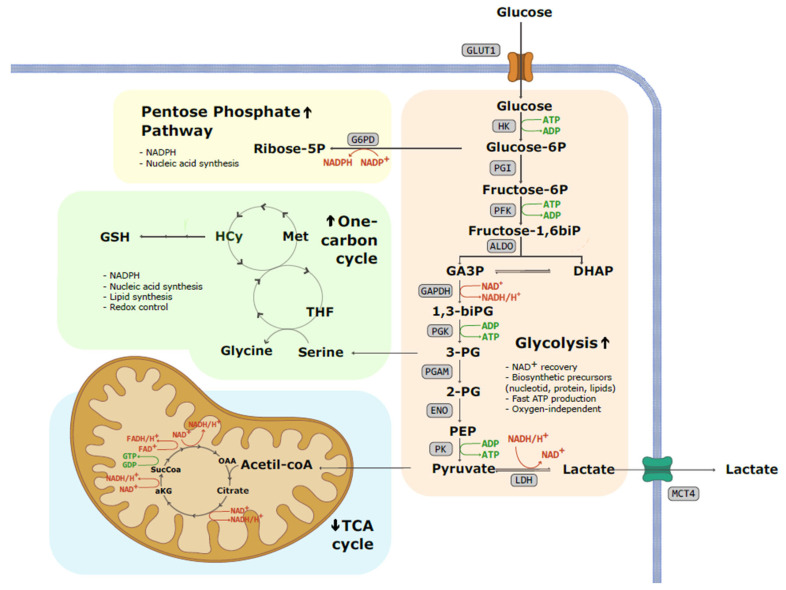
Cancer metabolism
Tumor cells don't just carry genetic mutations. They reprogram their metabolism. The Warburg effect (preferential use of glycolysis even in oxygen-rich conditions) isn't new, but its clinical implications are still being worked out. A 2025 review in Signal Transduction and Targeted Therapy documents how mitochondrial reprogramming, redox balance shifts, and substrate flux changes aren't passive byproducts of cancer growth. They're strategic, and they're directly tied to therapy resistance. Drugs that target metabolism rather than specific oncoproteins have had uneven clinical results, but the underlying biology is well-documented and actively studied.
Neurodegeneration and the brain's energy bill
The brain is metabolically expensive. At roughly 2% of body mass, it consumes close to 20% of total energy at rest. When brain energy metabolism is impaired, the consequences are serious.
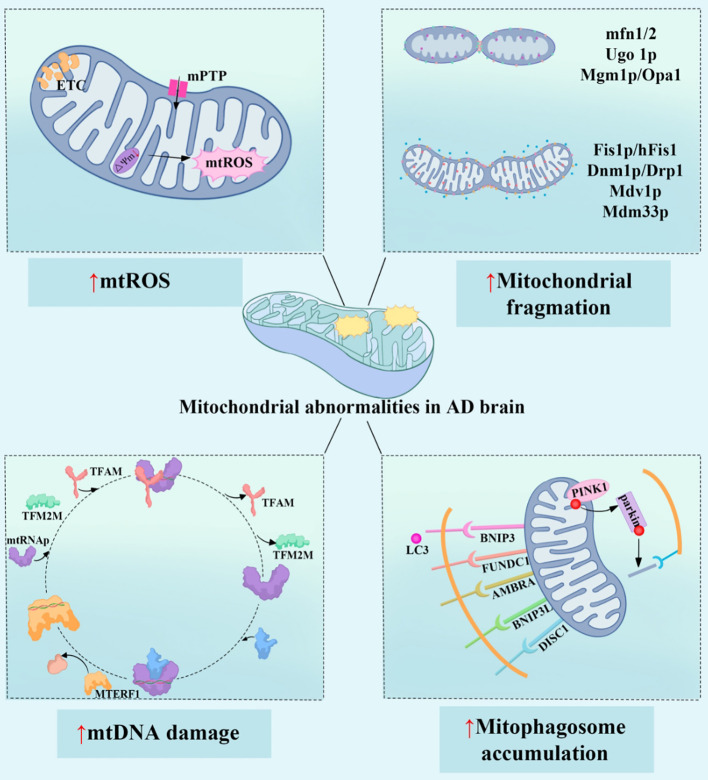
In Alzheimer's disease, glucose hypometabolism in certain cortical regions precedes the clinical onset of dementia by years. Mitochondrial dysfunction, impaired oxidative phosphorylation, and disrupted one-carbon metabolism are all documented features of Alzheimer's pathology. A 2025 analysis in Frontiers in Immunology identified four converging mechanisms: excess reactive oxygen species from mitochondria, dysregulated fusion and fission dynamics, mitochondrial DNA damage, and impaired mitophagy, each representing a distinct failure in the brain's energy maintenance system. The authors identified mitochondrial dysfunction as a key frontier for targeted therapies in neurodegeneration, while acknowledging that correcting these deficits in a whole organism with decades of adaptive compensation is genuinely hard.
Cerebrospinal fluid (CSF) remains the primary biospecimen for Alzheimer's biomarker research, and altered metabolite profiles in CSF appear well before cognitive symptoms emerge. The analytical challenge is significant: CSF is a low-volume specimen with complex composition, and the metabolites researchers are tracking (lactate, glucose utilization markers, mitochondrial dysfunction byproducts, one-carbon metabolites) are present in low concentrations with high inter-individual variability. Validating assays that can detect those signals reliably requires a well-characterized reference matrix that closely approximates the biological environment. For labs working in this space, that choice of matrix isn't a peripheral concern. It determines what you can trust in your data.

Why This Framing Matters Now
Target-centric drug development has produced real wins: GLP-1 agonists, certain KRAS inhibitors, and PCSK9 inhibition all work. But many diseases, especially those involving aging, neurodegeneration, chronic inflammation, and metabolic dysfunction, have resisted the single-target approach. The hits don't reliably stick.
One hypothesis is that we're solving the wrong abstraction layer. Identifying a disease-associated protein and inhibiting it doesn't account for what the body does with redirected energy or downstream allocation changes. A therapy that works on one axis may impose costs on another that weren't modeled.
Immunometabolism became a mature subfield precisely because the immune system's energy demands turned out to matter clinically. A 2024 review in Signal Transduction and Targeted Therapy documented how metabolic reprogramming in immune cells governs their function across health and disease. The insight that immune cells have specific metabolic requirements, and that those requirements shift with activation state, has opened new therapeutic angles that purely receptor-focused biology missed.
What an Energy Lens Could Add
This doesn't require abandoning molecular medicine; it requires adding a layer. Some concrete possibilities:
- Better research questions: What does this pathway cost energetically, and what does less of that resource mean for adjacent functions? These questions aren't routinely asked.
- Patient stratification: Patients with compromised metabolic capacity, including those with mitochondrial disease, type 2 diabetes, cachexia, or advanced age, may respond differently to metabolically demanding therapies than traditional biomarker-based stratification predicts.
- Side-effect modeling: Treatment fatigue, appetite loss, and behavioral changes may be interpretable as energy trade-offs rather than idiosyncratic toxicities. That reframing changes how you study and address them.
- Recovery planning: Healing has an energy cost. Post-surgical recovery, infection clearance, and cancer treatment all draw from the same finite budget. Modeling recovery as a resource allocation problem may produce better predictions than modeling it purely as tissue repair.
The Limits of the Argument
It's worth being direct about the limits here. The Nature essay is a conceptual argument, not a dataset. The authors explicitly frame it as encouragement to debate, not a clinical recommendation.
Energy is also notoriously difficult to measure cleanly in vivo. Whole-body expenditure, tissue-level metabolism, cellular ATP demand, mitochondrial state, and substrate flux are related but not interchangeable. Without operationalizable variables at the right scale, an energy framework risks remaining rhetorically compelling but clinically thin.
The history of mitochondria-targeting drugs is also humbling. Many have looked promising in preclinical models and disappointed in humans. That's not because the energy deficit is fictional; it's because correcting it through a drug in a whole organism is a different problem than demonstrating it exists.
Not every disease is best explained energetically, either. Some are dominated by very specific molecular lesions, structural damage, or toxic exposures where energy effects are downstream, not upstream. The framework is additive, not universal.
Where This Leaves Us
The body runs chemistry under budget constraints. That's not a new idea in evolutionary biology, and it's not new in metabolic ecology. What's potentially different is applying that framing systematically across biomedicine, treating energy not as background noise but as a variable worth measuring, modeling, and reasoning about explicitly.
Whether that pays off clinically depends on developing measurement tools that are robust enough to use in patients. The cases where the energy lens clearly adds something (sickness behavior, tumor metabolism, brain energy failure in neurodegeneration) suggest the phenomena are real. The question is whether the field can make the accounting precise enough to act on it.
Some of the most durable advances in medicine have come from frameworks that cut across disease categories. Energy constraints may not be the next one. But the argument that we've been systematically under-modeling them is hard to dismiss.
References
- Pontzer H. (2015). Constrained total energy expenditure and the evolutionary biology of energy balance. Exercise and Sport Sciences Reviews, 43(3), 114-120. PubMed
- Pontzer H. et al. (2021). Daily energy expenditure through the human life course. Science, 373(6556), 808-812. PubMed
- Brown JH et al. (2004). Toward a metabolic theory of ecology. Ecology, 85(7), 1771-1789. ESA Journals
- Wang A. et al. (2024). The immunology of sickness metabolism. Cellular & Molecular Immunology. Nature
- Signal Transduction and Targeted Therapy (2024). Metabolic regulation of the immune system in health and diseases: mechanisms and interventions. Nature
- Signal Transduction and Targeted Therapy (2025). Mitochondrial metabolism and cancer therapeutic innovation. Nature
- Wang S et al. (2025). Mitochondrial dysfunction in Alzheimer's disease: a key frontier for future targeted therapies. Frontiers in Immunology, 15:1484373. PMC
- Barba I, Carrillo-Bosch L, Seoane J. (2024). Targeting the Warburg Effect in Cancer: Where Do We Stand? International Journal of Molecular Sciences, 25(6):3142. PMC
Have questions about this research or our products?
Contact Our TeamGet new posts delivered to your inbox